ONE OLIGO : INFINITE POSSIBILITIES
Application Areas
A key difference between our synthesis technology and other next-generation platforms is the breadth of applications. Our technology offers higher quality and more flexibility to meet the growing number of new research, medical, and science areas. Below is a list of application areas that align closely with the advantages of our new oligos.
Order Express Oligos l Order EXTREmer long oligos l Request quote
Comparison
OTHER TECHNOLOGY: NICHE APPLICATIONS
On one hand, emerging chip-based synthesizers are good for applications that require enormous volumes of oligos, such as synthetic biology and NGS arrays. However, it is not well suited for other application areas due to the extreme volume and quality issues, which require additional enzymatic processing. Other issues include downstream processing and higher costs.
OUR TECHNOLOGY: ENDLESS APPLICATIONS
Eurofins Genomics’ next generation synthesizer is optimized for a much wider range of applications. Our oligos are chemically synthesized in individual solid supports that are ready-for-use when delivered salt-free after cleavage from the support. It offers a balance between high quality control and scalability which is perfect for PCR experiments. Since PCR is the backbone for most modern research—everything from medicine to the food you eat—the impact is massive.
OUR VISION
IMPACTS YOUR LAB
Our vision for this synthesizer technology is to support the scientific community and lower the barriers to their work, so you can do what you do best—better the human condition through science.
Our vision for this synthesizer technology is to support the scientific community and lower the barriers to their work, so you can do what you do best—better the human condition through science.
Scientists are constrained by resources, time, and money. These constraints impede medical, social and economic progress. What could have been the next big breakthrough for cancer treatment might be canceled because of budgets, bandwidth, or bureaucracy. At Eurofins Genomics, we believe that lowering the barriers between researchers and their work leads to more frequent breakthroughs that will change the world.
 |
 |
 |
|
HIGH FIDELITY
Unlike other “next generation” technologies, the quality on our oligos is outstanding.
|
FASTEST OLIGOS IN THE WORLD
Our vision for the finished product is to provide the fastest turnaround time in the industry.
|
LOWER PRICE
This lower scale platform provides cost advantages that we can pass along to our customers.
|
THEY PROMISED.
WE DELIVERED.
Everyone claims to have built the next generation synthesis technology, but many fall below expectations due to low quality or downstream issues. Eurofins Genomics US successfully engineered a next-generation DNA synthesizer that produces high quality, lower yield oligos in a shorter time. Also due to the incredible power and speed of the machine, it raises the ceiling on scalability and volume.
At Eurofins Genomics, we believe in proven science, not big promises. While other companies courted investor and media attention, we kept quiet and focused on the science. After years of diligent, hard work, we are proud of this new synthesis technology. We believe it will usher in a new era of research and scientific progress.
 |
 |
 |
|
SCALABILITY TO LARGE VOLUMES
The capacity for this technology is almost limitless, providing your greater flexibility to increase volume.
|
PCR PERFECT
High-fidelity, low-yield oligos that are prefect for any PCR based application.
|
OPTIMIZED OLIGOS
This new echelon of oligonucleotides are optimized for a variety of applications such as Genes, CRISPRs, and more.
|
ROCKET SCIENCE
BEHIND EVERY OLIGO
The initial concept for this technology was inspired by aerospace engineering on planes, almost 4 years ago. Building a DNA synthesizer is almost like building a small jet engine. Both utilize the science of air pressure, air flow, vibration, fluidics, and thermodynamics inside of complex, mechanical engineering.

Most companies would have partnered with a lab automation company. However, these companies have a higher tolerance for errors than commercial engineers. For instance, it is commonplace for lab equipment to be down for maintenance. Most scientists have experienced this personally in their lab. It happens so often that downtime is almost an accepted, social norm in the life science industry. We rejected the assumption that lab equipment cannot be both sophisticated and robust. We decided to take the path less traveled.
In aerospace engineering, downtime is unacceptable. Lives are at stake. For this reason, Eurofins Genomics partnered with a commercial-grade, aerospace engineering company to capture the same level of reliability and reproducibility found in airline industry.
- Reliable—consistent, controlled, and precise reagent flow ensure low QC failures
- Robust—phosphoramidite chemistry ensures >99.5% coupling efficiencies
- Rapid–increase in reagent dispensing and solvent removal speeds
ROBUST CHEMISTRY
MEETS ROBUST ENGINEERING
Innovation is born from hybridized ideas. DNA is no different. We combined the reliability and reproducibility of the aerospace engineering industry with the proven scientific method of phosphoramidite chemistry. The result is a robust, scalable synthesis process with over 99.5% coupling efficiency. Scalability comes from the high-throughput reagent handling and dispensing platform. It synthesizes the right amount of DNA sequences for you like your life depended on it.
|
|
The highly precise and controlled flow of microliter volumes of reagents, used in this synthesis platform, improves the oligonucleotides in several ways.
- Scalability—easily synthesize high volumes of oligos
- Small scale—between 1 and 2 nmole of oligos are produced
- Greener—reduction in the volume of chemical reagents
- Versatile—can synthesis short (15–60 nt) or long (60–200 nt) oligos
|
(One example of mass spectrometry from the new synthesis technology.)
BIG IDEAS
THAT TRANSCEND
Traditional high-throughput oligonucleotide synthesizers use a lot of reagents and make more DNA than that required in most applications. By contrast, emerging microarray or chip based methods generate too little DNA that, besides requiring additional downstream work before being useful, are only suitable for specific applications, predominantly, gene synthesis. As a result, researchers ordering DNA continue to pay for large quantities while requiring less than 1% of the same.
The capabilities of traditional liquid handling system used for high-throughput synthesis, however, are limited. Low volumes of reagent, microliters to sub-microliters, are not delivered efficiently in these systems, resulting in low oligo quality. To develop a system that delivered microliters of reagent solutions consistently and in a controlled manner, our scientist reached across the aisle to the experts in aeronautical engineering.
Eurofins Genomics started in 1986 as Operon Biotechnologies. Operon was the pioneer in high-throughput oligonucleotide synthesis, making synthesized DNA a reality for many scientist. 30 years later, Eurofins Genomics is still developing cutting-edge synthesizer technology to support science. You might say innovation is in our DNA.
Custom Oligo Technology
Eurofins MWG Operon uses optimized phosphoramidite chemistry and reagents of the highest quality. Proprietary synthesis platforms provide computer-controlled oligo synthesis, cleavage, deprotection, and desalting in a parallel processing format.
Eurofins MWG Operon oligonucleotides are synthesized with a high coupling efficiency (typically >99%) under salt-free conditions, avoiding the need for additional purification for most molecular biology applications, such as PCR, RT-PCR, sequencing, and hybridization studies.
The high coupling efficiency of Eurofins MWG Operon's proprietary synthesis technologies allows efficient synthesis of longmers up to 100 bases at high yields. In addition, a large number of modifications are available. All oligos are subject to stringent quality control (QC).
Synthesis Cycle
The synthesis takes place on a solid support -- controlled pore glass (CPG) -- where the 3'-nucleoside of the oligo being synthesized is attached. The oligonucleotide synthesis starts with the 3’ base. During the synthesis cycle the oligonucleotide is elongated toward the 5’ end.
For each coupling step, the following nucleotide is delivered as a nucleoside phosphoramidite where a reactive phosphoramidite group is located at the 3’-OH and the 5’-OH is modified with a dimethoxytrityl protection group (DMT).
The reactive phosphoramidite group reacts with the 5’-OH of the attached oligonucleotide.
The oligonucleotide synthesis cycle comprises the following steps:
- Detritylation:
The cleavage of the DMT protecting group from the previous base to form a 5’ reactive hydroxyl function.
- Coupling:
The 5’-OH group reacts with added activated phosphoramidite bearing the next base. As a result, both nucleosides are linked together.
- Capping:
The yield of the coupling step is - as every chemical reaction - less than 100%. Therefore any free 5’-OH groups which did not couple to the next nucleotide have to be excluded from the next coupling steps. Acetylation is used in the capping step to block all reactive 5’-OH groups.
- Oxidation:
The nucleotides are linked via phosphorous containing bonds that have been created in the coupling step. The phosphorus group is oxidized using iodine solution.
After the oxidation step a new synthesis cycle starts to add the next nucleotide (see step 1). The cycle is repeated until the desired sequence is synthesized.
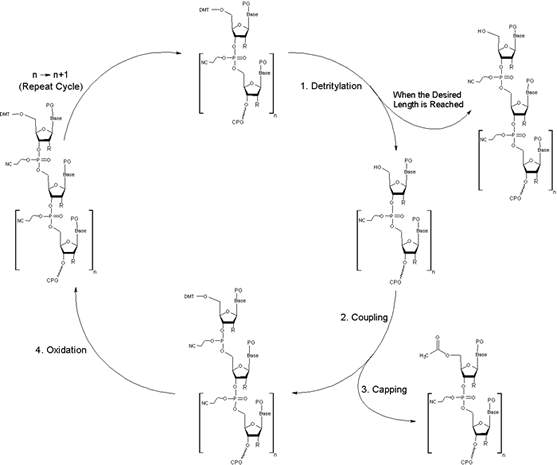
PG=Protecting Group; R depends on the type of nucleic acid being synthesized.
Click on image for larger view.
Deprotection:
After the synthesis has been performed and the desired length has been reached, the oligo undergoes one last detritylation reaction. The oligo is then cleaved from the solid support and the remaining protecting groups are cleaved to yield a biologically functional oligonucleotide.
How Modifications are Attached to an Oligo
Oligos can be modified in several different ways by utilizing the active groups of the nucleotide or creating nucleotide analogues. The most common oligo modifications are listed next:
- Terminal modifications utilizing the 3' and 5' OH groups
(e.g., C6 and C7 amino modifiers, biotin-ON, biotin-TEG, cholesterol-TEG, fluorescein, thiol modifications, phosphate)
- Base modifications
(e.g., 5-bromo-dU, 5-bromo-dC, 5-fluoro-dU, deoxyinosine, 5-iodo-dC, 5-iododU, 5-methyl-dC, 5-nitroindole, deoxyuridine)
- Thymidine analogues, replacing a T residue in the sequence
(e.g., C2 dT and C6 dT amino modifiers, biotin-dT, dabcyl-dT, fluorescein-dT, TAMRA-dT)
All the previously listed modifications are available internally and on the 5' end. Some are also available on the 3' end. Most 3' modifications require special controlled pore glass (CPG) columns, which are not available for all modifications.
- Post-synthesis modifications
Modifications are attached post-synthetically, via an amino modifier (e.g., coumarin, digoxygenin, Oregon Green®, ROX, Texas Red®-X, BODIPYdyes). By choosing the appropriate amino modifier, these modifications can be attached in different positions in the oligo.
- Modifications of the phosphate group
(e.g., phosphorothioation)
- 2' Modifications
(2'-O-methyl A/C/G/U, ribo A/C/G/U)
NOTE: Unmodified oligos are very stable molecules; however, with the addition of modifications, their properties can change significantly. Modified oligos are more sensitive to light, pH, and frequent freezing and thawing. Proper storage and handling of modified oligos is imperative.